Metformin: From Foundational Diabetes Therapy to Longevity Contender
Metformin is a first-line therapy for type 2 diabetes. Type 2 diabetes is a disease where the body does not respond to the insulin trigger to reduce blood glucose levels and so untreated Type 2 diabetics will experience dangerously high blood sugar levels.
Metformin has been in use for diabetics for centuries starting with a herbal plant remedy used to treat “sweet urine” disease and it was studied, refined, and manufactured into a pharmaceutical beginning in the 1920s. While its clinical role in the use of glucose regulation is well known, there is growing clinical interest in its role in aging science.
Traditional uses of Metformin
Diabetes
Metformin is a first-line, gold standard therapy in the treatment of type 2 diabetes mellitus. Its mechanism is primarily understood as a reduction of the liver’s release of stored glucose, increasing insulin sensitivity, and decreasing intestinal glucose absorption. In practice, metformin is shown to lower diabetic A1c by 1-1.5% with minimal risk of lowering the blood sugar too far. If changes in weight occur, it would be mild weight loss but we do not see weight gain which is important in diabetics.
Metformin overall is highly effective and safe, while being widely available as a low-cost generic.
Prediabetes and Insulin Resistance
Metformin is also used in patients that are at high risk for diabetes or are pre-diabetic. These are patients with high BMI (≥35), under 60, and women with a history of gestational diabetes. Using metformin in these cases can support the body and delay diabetes disease progression.
Polycystic Ovary Syndrome (PCOS)
PCOS is a common hormonal disorder in women that causes irregular periods, infertility, and excess androgen hormones. PCOS is marked by insulin resistance and so metformin is used to help regulate the body’s insulin sensitivity. We can also see patient’s ovulatory function and androgen levels improve with metformin treatment. It is often used alongside fertility treatments to improve reproductive outcomes.
Weight loss and Metabolic Syndrome
Metformin can be used off-label as a weight management therapy to yield mild weight reduction through visceral fat elimination. We also see an improvement in patient lipid profiles with a reduction low-density lipoprotein (LDL -10%) and triglyceride (TG -10%), these can be accompanied by an increase in high-density lipoprotein (HDL +7%).
Other Clinical Uses
Other clinical and off-label applications include:
Non-alcoholic fatty liver disease (mixed evidence)
Gestational diabetes (select cases)
Antipsychotic-induced weight gain
Metformin Mechanism and Relation to Longevity Science
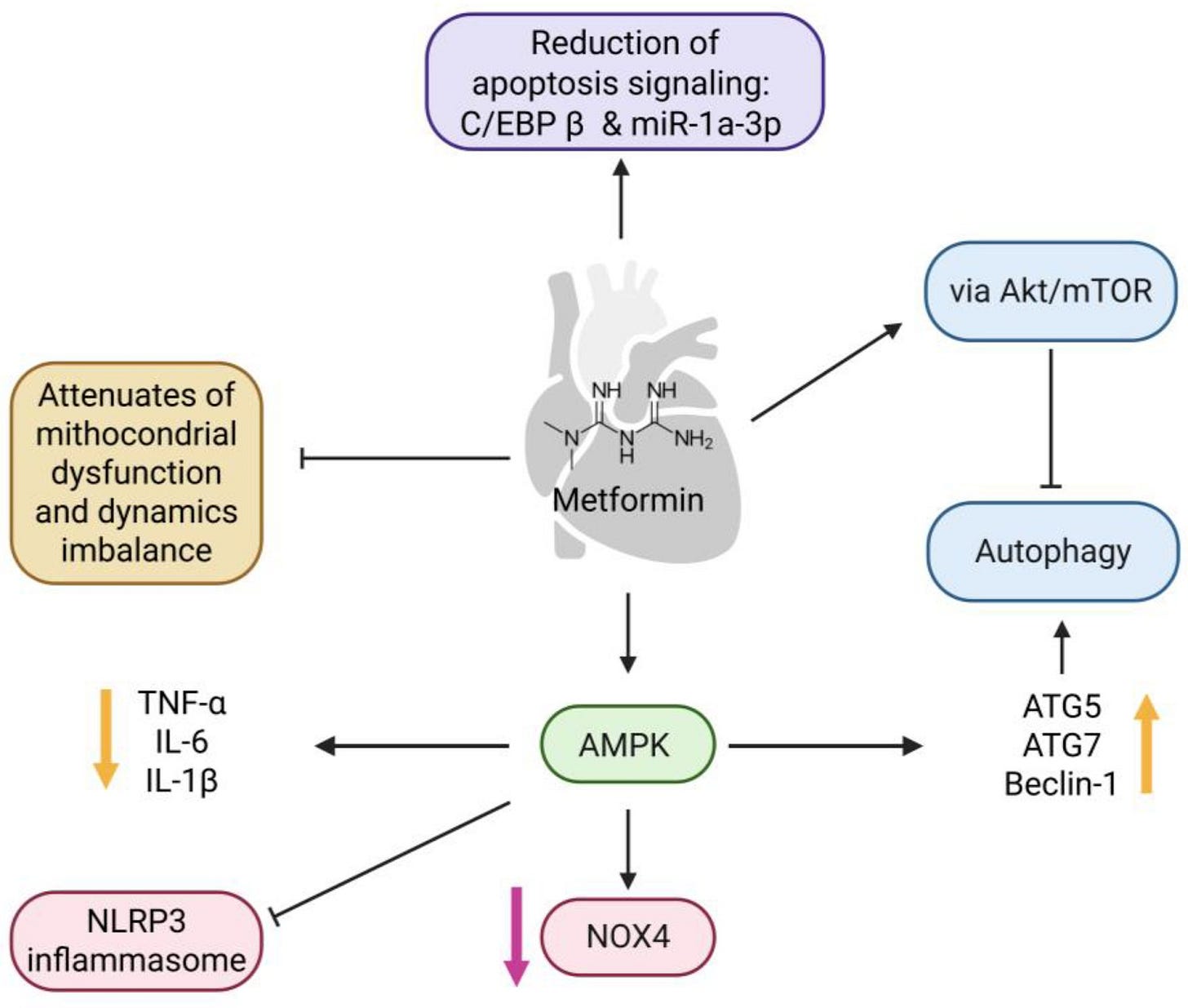
Metformin’s transition into longevity science stems from its effects on key biological pathways associated with aging.
Its primary actions include:
Reducing liver glucose production via gluconeogenesis
Increasing glucose uptake through activating AMPK signalling and inhibiting mTOR
Reduction in mitochondrial reactive oxygen species (ROS)
Enhancement of autophagy
Modulation of the gut microbiome
Collectively, these effects mimic fasting physiology.
At a cellular level, metformin inhibits mitochondrial electron transport (specifically, complex I), activates AMPK, suppresses mTOR, and promotes autophagy and mitochondrial biogenesis. Beyond AMPK activation, metformin influences cell life cycle, chronic inflammation via NFκB inhibition, and may delay stem cell aging.
Metformin as a longevity drug remains unproven and controversial though mechanistically it may look attractive especially when combined with data from diabetes trials. Current evidence does not support metformin for healthy people outside of clinical trials or diabetes prevention.
Preclinical and Animal Longevity Data
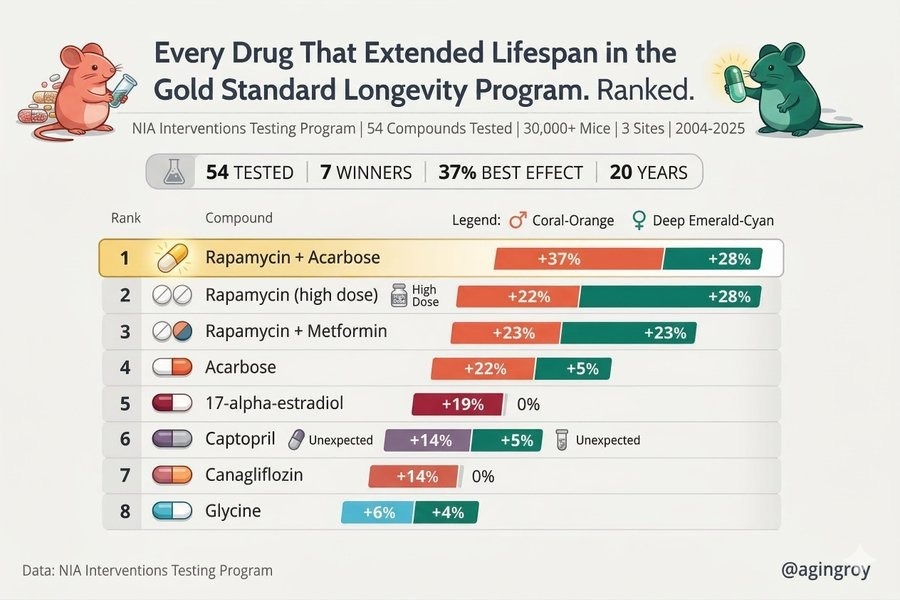
Animal studies have produced mixed results. While metformin extended lifespan in some organisms such as C. elegans, it failed to extend lifespan in mice when used alone. However, combination therapy with rapamycin demonstrated significant lifespan extension.
A primate study in macaque monkeys showed metformin preserved memory, reduced cortical thinning, maintained muscle fibers, and reduced cellular senescence markers over 40 months.
Observational Human Data
Studies show that diabetic patients have reduced all cause mortality rates when taking metformin. Observational studies also show that diabetic patients taking metformin may experience equal or lower mortality rates compared to non-diabetics. The reduced mortality finding goes along with reduced incidence of cancer, cardiovascular disease, and possibly cognitive decline.
Cardiovascular Protection
The landmark UK Prospective Diabetes Study (UKPDS) followed a 20 year timeline of 5,000 Type 2 diabetic patients showing early, effective disease management using metformin reduces microvascular complications and reduced cardiovascular mortality. Specifically overweight patients with newly diagnosed type 2 diabetes being treated with metformin showed reduced instances of myocardial infarction by 39%, coronary deaths by 50%, stroke by 41%, and all-cause mortality by 36% over 10.7 years compared to diet alone. These reductions were greater than those achieved with sulfonylureas or insulin.
A 2021 meta-analysis of randomized controlled trials found metformin associated with a 48% reduction in major adverse cardiovascular events and a trend toward lower all-cause mortality. Recent 2025 cohort data suggests metformin’s protective effects on cardiovascular events and mortality persist for approximately 8-10 years after treatment initiation.
This cardiovascular protective effect may be limited however as a recent study published this month from researchers out of Rutgers University found that exercise when combined with metformin use offered decreased benefits including no increase in VO2 max with continued exercise, blunted effect on inflammation, and no exercise-initiated metabolic benefits to insulin and fasting glucose.
Anti-Cancer Potential
Metformin has been associated with reduced rates of several cancers, including breast, colon, and prostate cancer. Proposed mechanisms include decreased insulin/IGF-1 signaling and reduced metabolic fuel availability for tumor growth.
Neuroprotection
Potential neuroprotective effects include reduced neuroinflammation and improved mitochondrial efficiency. While data in Alzheimer’s disease remains mixed, there is some indication of benefit in early disease stages.
Clinical Trials for Metformin and Aging
The Targeting Aging with Metformin (TAME) is an approved clinical trial designed to test whether a single agent, metformin, can slow age related disease progression in older adults without diabetes. The goal would be to see if using metformin can delay the onset of age-related conditions such as cancer, cardiovascular disease, cognitive decline, and mortality. Although the trial received regulatory approval over ten years ago and the medical community at large is highly interested, the trial has still yet to take off due to various reasons including funding. As metformin is a low-cost, generic pharmaceutical, pharmaceutical companies do not have a big enough incentive to support the huge cost for clinical trials. Currently the trial is seeking private funding sources.
Data emerging from the smaller MILES (Metformin In Longevity Study) trial suggest metformin may induce anti-aging transcriptional changes in older adults with impaired glucose tolerance (but not full diabetes). The transcriptional changes alter gene expression for pathways associated with metabolic function (PCK1, SDHC, CETP) and cell cycle/cell death pathways (E2F1, PARP1, RET).
Practical Use in Longevity Medicine
Typical Off-Label Protocols
In longevity-focused settings, metformin is often used at doses of 500–1500 mg per day, typically in extended-release form.
It is most commonly considered in:
Insulin-resistant, non-diabetic individuals
High risk longevity populations with family history of diabetes and cardiovascular disease
Elevated fasting insulin or HOMA-IR
Sedentary or overfed metabolic states
High cardiometabolic risk profiles
Who May Not Benefit
Metformin may offer limited or even counterproductive effects in:
Lean, insulin-sensitive individuals
High-performance athletes
In athlete populations, metformin may blunt mitochondrial adaptations to exercise and reduce improvements in VO2 max and muscle hypertrophy.
Risks, Downsides, and Controversies
Mitochondrial Effects: inhibition of mitochondrial complex I has contradictory outcomes
Reduces oxidative stress
Limits peak energy output
Exercise Adaptation: reduced VO2 max gains and blunted muscle growth
Nutrient Deficiencies: commonly associated with B12 deficiency and possible interference with folate
Gastrointestinal Side Effects: nausea and diarrhea (esp with immediate-release dose)
References:
doi:10.2337/dc26-S009.
doi:10.1007/s00125-017-4336-x.
doi:10.7326/M15-2650.
doi:10.1016/j.jacc.2020.02.056.
doi:10.1161/CIR.0000000000000766.
doi:10.1016/j.numecd.2020.11.031.
doi:10.1016/j.diabres.2019.107946.
doi:10.1038/s41598-025-89721-7.
doi:10.2337/dci22-0034.
doi:10.7326/M16-1860.
doi:10.1016/j.jacc.2021.02.057.
doi:10.1016/j.jacc.2023.05.038.
doi:10.1016/S2213-8587(23)00001-3.
doi:10.3389/fendo.2021.718942.
doi:10.1016/j.jacc.2023.05.038.
doi:10.1001/jama.2025.11289.
doi:10.1016/j.cmet.2020.04.001.